Lifetime Citizen Portal Access — AI Briefings, Alerts & Unlimited Follows
Lawmakers press NIH on grant pauses, terminations and clinical‑trial impacts; appeals process established
Loading...
Summary
Senators from both parties pressed NIH Director Jay Bhattacharya about thousands of paused or terminated grants, delayed award notices and clinical trials affected; Bhattacharya said the agency has set up an appeals process and will review terminations, while senators demanded detailed counts and timelines for restoration.
Lawmakers repeatedly questioned NIH Director Jay Bhattacharya about the agency’s recent pauses, terminations and delays of grant awards and the downstream effects on clinical trials and patient care. Several senators said thousands of grants have been affected and cited specific dollar amounts; Bhattacharya said the agency has opened an appeals process and was working to restore appropriate funding.
What lawmakers said
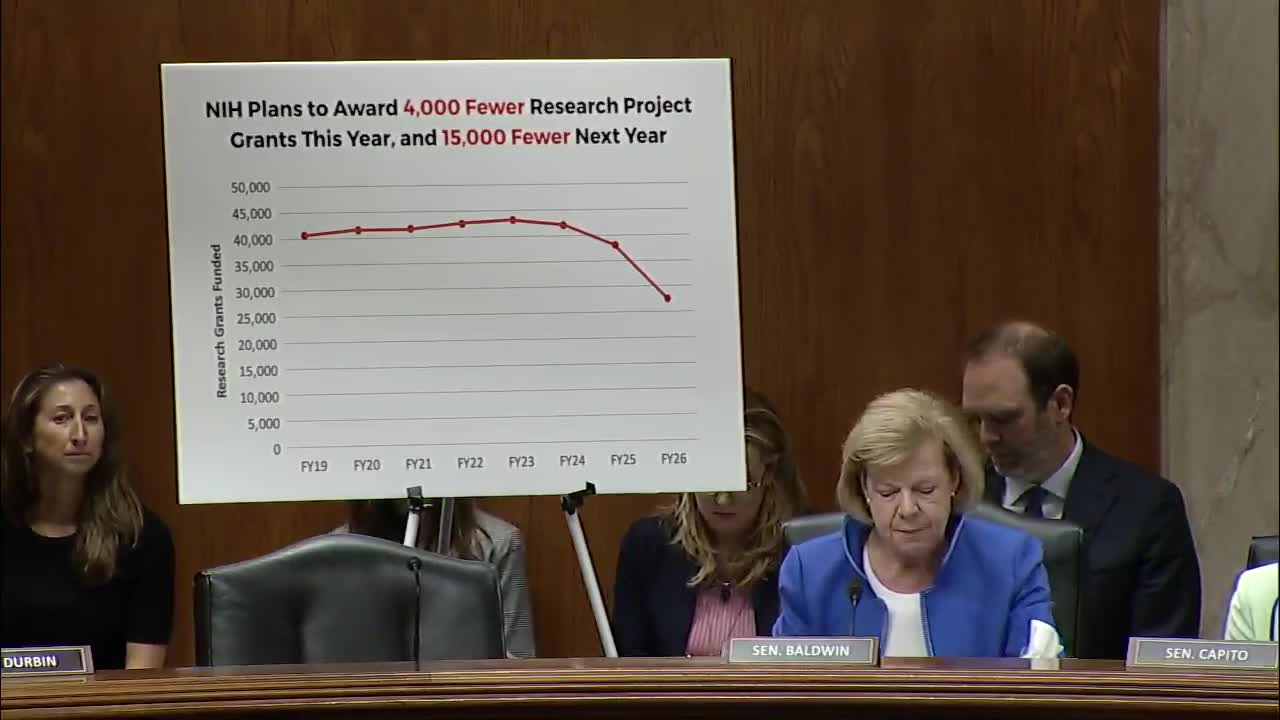
• Counts cited by senators: Senator Tammy Baldwin said NIH had “terminated 2,370 active grants and $4,900,000,000 in funding.” Senator Murray testified to the committee that the administration had “terminated nearly 2,500 grants totaling almost $5,000,000,000” and prevented nearly $3,000,000,000 in grant funding from being awarded. Members also said NIH had awarded roughly $3,000,000,000 less so far this year and that 3,288 fewer grants had been awarded through the same period as last year.
• Clinical trials impact: Senators, including Murray and Durbin, told the director that at least 160 clinical trials had been terminated or otherwise impacted, and raised examples including an HIV vaccine research network and cancer and diabetes trials that senators said were interrupted.
Director’s response and appeals process
Bhattacharya said he established a process for appeals of pauses and terminations, that hundreds of appeals have been submitted, and that many decisions have been or will be reversed following review. He said he did not take the job to terminate grants and emphasized his commitment to making sure allocated funds support research that advances health needs. When asked who was making pause/termination decisions, he said the answer varies by case and that some pauses related to settlement negotiations with institutions predated his tenure; other decisions reflected policy changes instituted by the administration.
Unresolved requests and committee follow‑up
• Senators demanded detailed, itemized counts by institute, center and job function of staff reductions and grant impacts. Senator Murray requested a full accounting of staff reductions and of clinical trials affected; Bhattacharya said some numbers were in flux and that he would provide additional information to the committee.
• Timing: Senators sought a firm timeline for processing appeals. Bhattacharya said appeals would not take “years” and indicated the agency is processing them on a timeline of “weeks,” and Senator Schatz said he would follow up frequently.
Ending note
Committee members warned that rapid pauses and broad keyword‑based reviews earlier in the administration had swept up many unrelated projects. They pressed Bhattacharya to ensure the appeals process is timely and transparent and to provide the detailed counts and the list of clinical trials affected so Congress can assess appropriation and oversight options.