Presenter says portable combined tests could cut forensic backlogs but standards and workflows limit gains
Get AI-powered insights, summaries, and transcripts
Subscribe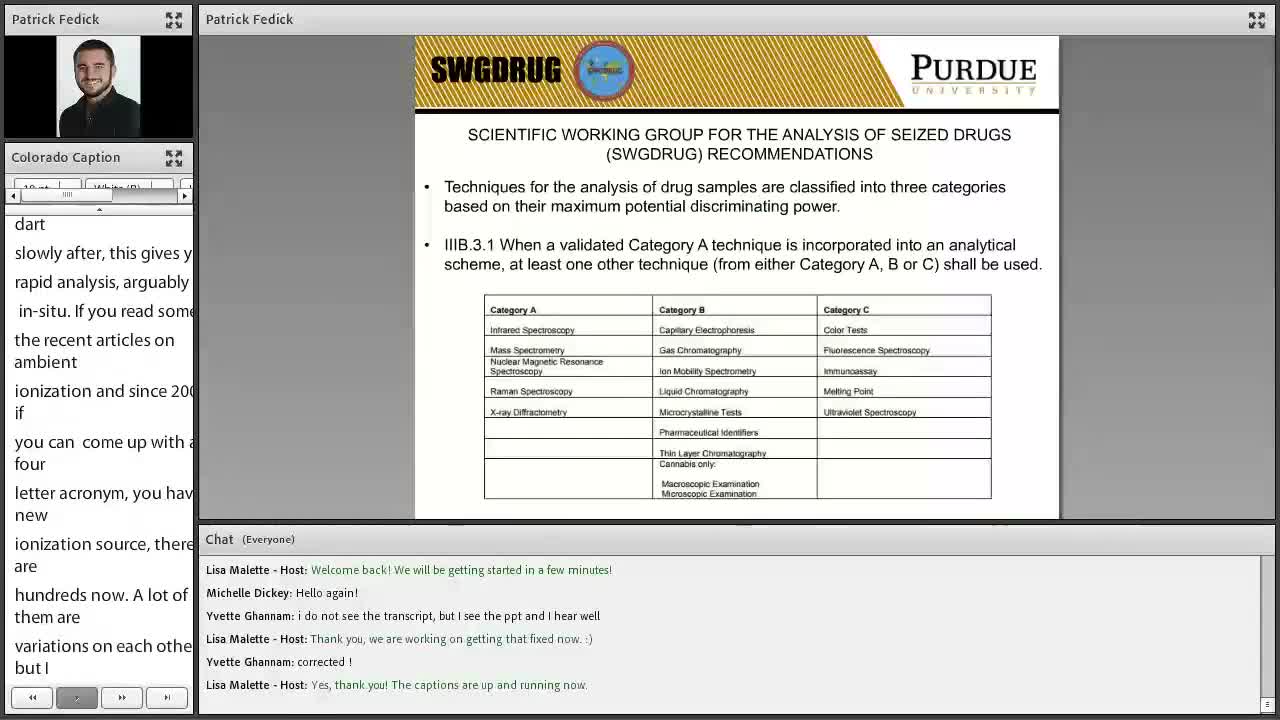
Summary
The presenter argued portable Raman plus paper-spray mass spectrometry could reduce forensic lab backlogs and speed roadside screening, but cited Scientific Working Group requirements and current lab practices as obstacles to realizing those time savings.
Unidentified Speaker, presenter, described operational implications of combining portable Raman spectroscopy with paper-spray mass spectrometry and warned that validation standards and existing lab workflows can blunt the time-saving benefits of rapid screening.
The presenter gave a hypothetical roadside scenario: an officer finds a small bag of suspected controlled substance and runs a paper‑spray screen that, in seconds, indicates cocaine. He said that under Scientific Working Group (SWG) practices, a confirmed identification typically requires an additional validated technique such as chromatography or a color test. "All that time that you saved on your ambient ionization was for nothing because you now have to go and do a second test," the presenter said, describing how confirmatory requirements can reintroduce delays.
Drawing from an internship at the Department of Homeland Security, Customs and Border Protection (CBP), the presenter described setting up a 40‑plus designer‑drug screening system and criticized the common practice of re‑running rapid screens through LC‑MS workflows. "You guys are wasting so much time," he said, framing this as an operational inefficiency that combined Raman/MS methods could help address if agencies adjust validation and workflow practices.
The presenter proposed two operational priorities to realize field benefits: building combined spectral databases to support on‑scene identifications and validating portable Raman plus MS workflows so results can be admissible without routine reanalysis in the central lab. He cautioned these are research and development steps — not yet policy changes — and recommended further testing and interagency coordination before operational adoption.
The presenter closed by thanking funders (DOE for his lab and DOJ for a collaborator's lab) and inviting questions.
